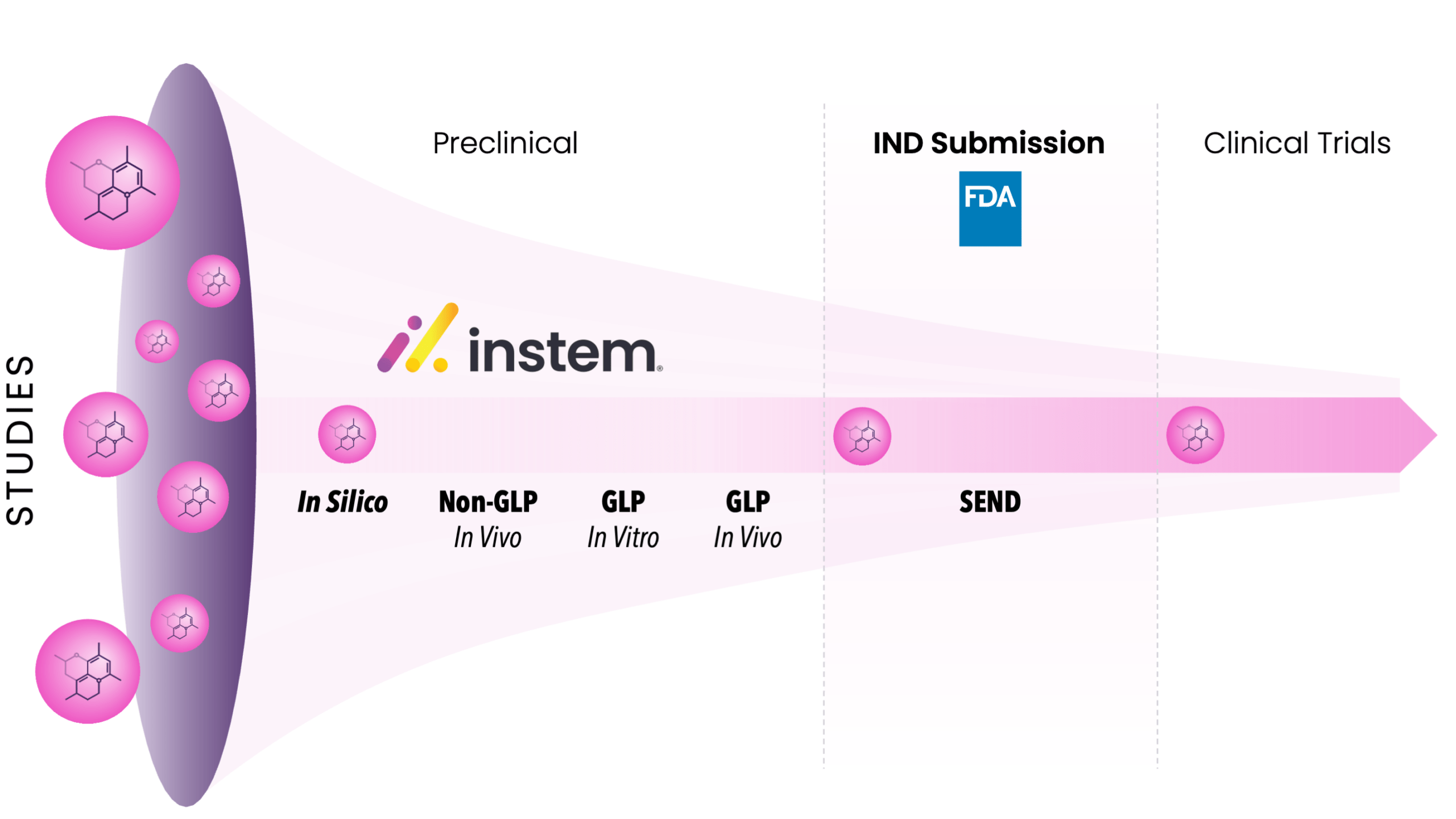
The earliest stages of drug research and development (R&D) generate massive volumes of scientific, operational, and compliance-critical data, often across disconnected tools, teams, and formats. That fragmentation slows decision-making, increases rework, and introduces quality and traceability risks precisely when teams need speed and confidence. This blog series explores how Instem’s portfolio is designed to connect the journey of drug discovery from end-to-end, providing robust support and compliance from target identification and toxicology to submission-ready Standard for Exchange of Non-Clinical Data (SEND) dataset creation and validation.
Instem Supports Each Stage of Drug Discovery
With advanced in silico toxicology, centralized study management, modern laboratory execution, and SEND-ready submission solutions, Instem is a single source provider of the tools needed to navigate the data path through preclinical to IND Submission. Our solutions help maintain data integrity across target identification, discovery, lead optimization, preclinical efficacy, toxicology, Chemistry, Manufacturing and Control (CMC) readiness, and regulatory submission. The result is faster, more confident decision-making, reduced developmental risk, and higher-quality, regulatory-ready data at every stage of drug discovery.
Target Identification
Target selection is one of the most crucial aspects in drug discovery. Teams must connect biological target plausibility to early safety signals, such as on-target liabilities, pathway-level risks, and adverse events, to identify and validate suitable targets. This can be challenging when there is a plethora of unstructured data scattered across multiple sources. Manually reviewing literature and toxicology datasets is time-consuming, and scattered data storage increases the likelihood of overlooking toxicological signals.
Fortunately, Instem provides a solution to these challenges through KnowledgeScan™ and Centrus®, platforms that support researchers with more efficient and robust target identification by providing a clearer view of biological rationale and potential toxicological risk.
Insightful Safety Assessments with KnowledgeScan
The KnowledgeScan platform is a robust Target Safety Assessment (TSA) service that ensures all relevant information is considered from the start of the target identification process.
Firstly, reliable, up-to-date safety assessments are built by extracting and refining millions of data points from published sources across pharmacology, bioinformatics, and toxicology. Through this, KnowledgeScan streamlines literature reviews, delivers an impartial and comprehensive view of vast amounts of data, and removes the risk of overlooking critical toxicological signals. Every report is curated and validated by scientific experts. Overall, this supports faster target selection and drug candidate profiling.
Centrus: Safety Intelligence in Drug Discovery
Centrus is a cutting-edge platform designed for organizations of all sizes interested in harnessing high-value datasets to improve safety assessments.
Where basic data storage and visualization tools may hinder informed decisions and safety outcomes, Centrus drives harmonization across a vast amount of data and is capable of intelligently linking clinical and preclinical data. This enables researchers to identify potential adverse events before clinical trials begin, and make more informed decisions backed by data.
The centralized platform allows collaboration between departments and organizations to occur seamlessly, helping to identify risks earlier. These features promote more efficient and successful target or candidate identification.
By enhancing visibility, strengthening safety assessments, and consolidating complex datasets, Instem gives teams the clarity and confidence needed to move forward with stronger targets and fewer risks. With KnowledgeScan and Centrus forming the backbone of early discovery intelligence, organizations can accelerate decision‑making and build a more robust foundation for the entire journey of a molecule. In the next post of our series, we explore how these insights feed seamlessly into the broader discovery process.


