About Us
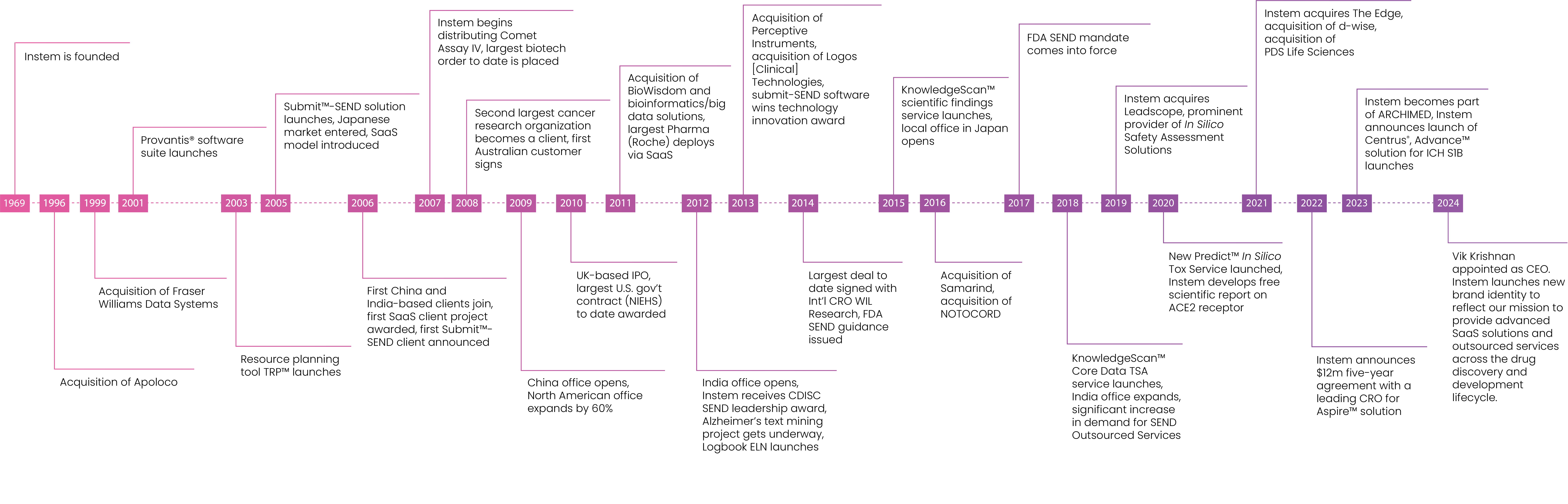
Established in 1969, we’ve been supporting complex research projects for over 50 years.
Our Vision
To help our customers discover, accelerate, and advance key R&D programs by offering solutions powered by science and best practices in research.
We’re committed to supporting our clients with innovative, leading SaaS platforms and services that empower scientific discovery, accelerate research studies, and advance human health.

Instem Worldwide
Founded in the United Kingdom in 1969, Instem has deep roots internationally across North America, EMEA and APAC.
We maintain a commercial and technical presence throughout these regions and pride ourselves on localized support for our diverse client base.
Industries
we serve
- $Pharmaceutical
- $CRO
- $CDMO
- $Academic Research
- $Biotechnology
- $Industrial
- $Agrichemical
More about us
Join us in transforming life sciences R&D—meet our leadership or explore careers that make a difference.
Meet the experienced leaders dedicated to advancing solutions for our clients’ success.
Empower Your Research
Start Your Transformation Today
